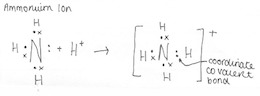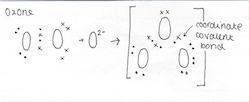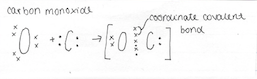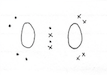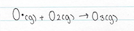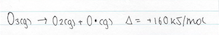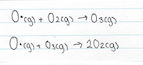Coordinate Covalent Molecule
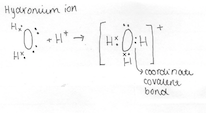
4.4 Describe the formation of a coordinate covalent bonds and illustrate them using Lewis electron dot structures
A covalent bond is an attractive force that is caused by two electrons being shared between two non-metal elements. A covalent bond is formed between two non-metals that have similar electronegativity’s. Neither atom is "strong" enough to attract electrons from the other. For stabilization, they share their electrons from outer molecular orbit with others. Neither atom is "strong" enough to attract electrons from the other. For stabilisation, they share their electrons from outer molecular orbit with others. A coordinate covalent bond is formed in an identical manner to an ordinary covalent bond and therefore aren’t different to an ordinary covalent bond.
A covalent bond is an attractive force that is caused by two electrons being shared between two non-metal elements. A covalent bond is formed between two non-metals that have similar electronegativity’s. Neither atom is "strong" enough to attract electrons from the other. For stabilization, they share their electrons from outer molecular orbit with others. Neither atom is "strong" enough to attract electrons from the other. For stabilisation, they share their electrons from outer molecular orbit with others. A coordinate covalent bond is formed in an identical manner to an ordinary covalent bond and therefore aren’t different to an ordinary covalent bond.
| Physical properties | Chemical properties | Example | |
| Single covalent bonds | Longest bond length in comparison to those below | One pair of electrons are shared | Hydrogen (H2) |
| Weakest covalent bond | |||
| Double covalent bonds | Average in length to those above and below | Two pairs of electrons are shared | Oxygen (O2) |
| Most average covalent bond | |||
| Triple covalent bonds | Shortest bond length in comparison to those above | Three pairs of electrons are shared | Nitrogen (N2) |
| Strongest covalent bond |
- - - - -
The oxygen molecule is a linear diatomic molecule, which shares a double covalent bond holding the two oxygen atoms together. The molecule is non-polar.
The oxygen molecule is a linear diatomic molecule, which shares a double covalent bond holding the two oxygen atoms together. The molecule is non-polar.
4.5 Demonstrate the formation of coordinate covalent bonds using Lewis electron dot structures
Allotropes of Oxygen
4.6 Compare the properties of the oxygen allotropes O2 and O3 and account for them on the basis of molecular structure and bonding
Allotropes are forms on one element (in the same physical state) that have distinctly different physical properties, including hardness, electrical conductivity and density. The most common allotropes of oygen are O2 (left) and O3 (right) illustrated below
Allotropes are forms on one element (in the same physical state) that have distinctly different physical properties, including hardness, electrical conductivity and density. The most common allotropes of oygen are O2 (left) and O3 (right) illustrated below
| Oxygen (O2) | Ozone (O3) | |
| State | Gas | Gas |
| Appearance | A colorless gas condensing to a pale blue liquid | A pale blue toxic gas condensing to a distinctly blue liquid |
| Odor | Odorless | A strong and distinctive sharp pungent odor (O.01ppm is detectable) |
| Boiling point | -182.96°C | -110.5°C |
| Density | At 20°C – 1.331g/L | At 20°C – 1.998g/L |
| Density is comparable to the density of air | Density, when compared to air, is 1.5 times | |
| Bonding | A diatomic molecule formed by photosynthesis and contains two oxygen atoms held together with a covalent, double bond | Formed by UV radiation and electric discharge on oxygen, it is composed by three oxygen atoms held together with a one single bond and one double bond. The single bonds I formed by two oxygen atoms sharing a pair of electrons, that both came from the one of the atoms (coordinate covalent bonding) |
| Shape | Linear molecular shape | Bent molecular shape |
Intramolecular bonds are those forces between the atoms found inside a single molecule. Examples of intramolecular bonds include metallic, ionic and covalent bonds. Intermolecular forces are those forces between molecules. These forces determine the boiling point of substances thus the state. (solid, liquid or gas). The stronger the IMF the higher the boiling point as more energy is needed to break the bonds. Examples of intermolecular bonds include dipole-dipole forced, dispersion forces and hydrogen bonds.
- - - - -
Both O2 and O3 are covalent molecular substances which result in them having low melting and boiling point and density. Covalent bonds between atoms are quite strong, but attractions between molecules/compounds, or intermolecular forces, can be relatively weak. Covalent compounds generally have low boiling and melting points and density, and are found in all three physical states at room temperature due to the presence of weak intermolecular forces.
- - - - -
The O3 molecule consists of three oxygen atoms, one single coordinate covalent bond and one double covalent bond. The two O-O that share the double covalent bond are nonpolar as there is no electronegativity between these atoms of the same element, sharing the same number of electrons. However, the distribution of the electrons along the 3 oxygen atoms is uneven, as the central atom must share electrons with the two atoms either side of it, whereas the outer atoms must share only with the central one. Due to the electrons being taken away (shared) from the central atom, this atom is more deprived of electrons then the other two-oxygen atoms. As a result of this sharing the central atom has a formal charge (a formal charge (FC) is the charge assigned to an atom in a molecule, assuming that electrons in a chemical bond are shared equally between atoms, regardless of relative electronegativity) of +1 while the other atoms each have a formal charge of -½. Since the molecule is bent in terms of geometry, the result of this sharing of electrons results in a dipole force across the entirety of the ozone molecule.
Ozone has a higher melting and boiling point and density than the oxygen molecule as the ozone molecule essentially has three bonds (one double and one single) and as a result more force in the form of heat, must be used to break all three of the bonds in the ozone molecule. Ozone contains a double bond, which will break apart easily, however once this double bond is removed, there remains two single bonds, which will require more energy to break. Whereas its cousin, molecular oxygen has only two bonds – one double, and is therefore reactive as this bond will readily break, however the breaking of the last bond needs a little more heat. The difference in density and boiling/melting points between ozone and molecular oxygen are due to the extra bond in Ozone, which is not present in oxygen. The below flow chart highlights the extra bond in ozone.
Ozone: Double and single bonds à two single bonds à one single bond à ozone free radical
Molecular oxygen: Double bond à single bond à ozone free radical
- - - - -
The majority of the differences in properties between ozone (O3) and oxygen (O2) can be explained in regards to their differing chemical structures. When dioxygen reacts, the double covalent bond between the two oxygen atoms in the molecule must be broken. The splitting of the double covalent bond between the oxygen’s require considerable amounts of energy
- - - - -
Both O2 and O3 are covalent molecular substances which result in them having low melting and boiling point and density. Covalent bonds between atoms are quite strong, but attractions between molecules/compounds, or intermolecular forces, can be relatively weak. Covalent compounds generally have low boiling and melting points and density, and are found in all three physical states at room temperature due to the presence of weak intermolecular forces.
- - - - -
The O3 molecule consists of three oxygen atoms, one single coordinate covalent bond and one double covalent bond. The two O-O that share the double covalent bond are nonpolar as there is no electronegativity between these atoms of the same element, sharing the same number of electrons. However, the distribution of the electrons along the 3 oxygen atoms is uneven, as the central atom must share electrons with the two atoms either side of it, whereas the outer atoms must share only with the central one. Due to the electrons being taken away (shared) from the central atom, this atom is more deprived of electrons then the other two-oxygen atoms. As a result of this sharing the central atom has a formal charge (a formal charge (FC) is the charge assigned to an atom in a molecule, assuming that electrons in a chemical bond are shared equally between atoms, regardless of relative electronegativity) of +1 while the other atoms each have a formal charge of -½. Since the molecule is bent in terms of geometry, the result of this sharing of electrons results in a dipole force across the entirety of the ozone molecule.
Ozone has a higher melting and boiling point and density than the oxygen molecule as the ozone molecule essentially has three bonds (one double and one single) and as a result more force in the form of heat, must be used to break all three of the bonds in the ozone molecule. Ozone contains a double bond, which will break apart easily, however once this double bond is removed, there remains two single bonds, which will require more energy to break. Whereas its cousin, molecular oxygen has only two bonds – one double, and is therefore reactive as this bond will readily break, however the breaking of the last bond needs a little more heat. The difference in density and boiling/melting points between ozone and molecular oxygen are due to the extra bond in Ozone, which is not present in oxygen. The below flow chart highlights the extra bond in ozone.
Ozone: Double and single bonds à two single bonds à one single bond à ozone free radical
Molecular oxygen: Double bond à single bond à ozone free radical
- - - - -
The majority of the differences in properties between ozone (O3) and oxygen (O2) can be explained in regards to their differing chemical structures. When dioxygen reacts, the double covalent bond between the two oxygen atoms in the molecule must be broken. The splitting of the double covalent bond between the oxygen’s require considerable amounts of energy
Ozone contains both a covalent double bond and a covalent single bond and as a result, when this ozone reacts the covalent single bond with break more readily, resulting in the spitting off of one Oxygen atom, leaving a stable O2 molecule behind. The energy to do this is much less than what is needed to break a double bond
As a result O3 is more reactive than O2 as less energy is required to break a covalent bond. Single coordinate covalent bonds are weaker than double bonds, and as ozone contains one of each, the single coordinate covalent bond will react with the least amount of energy. Oxygen (O2) contains only a double bond, requiring more energy to break than that of a single bond, resulting in ozone being more reactive.
Oxygen Free Radicals
4.7 Compare the properties of the gaseous form of oxygen and the oxygen free radical
A free radical is a neutral species which possesses an unpaired electron, and which can be formed by splitting a molecule into to neutral fragments.Free radicals are generally very reactive due to the presence if an unpaired electron, and an incomplete valence shell.
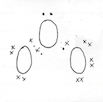
Three examples of free radicals are sown below, oxygen free radical, chlorine free radical and bromine free radical
A free radical is a neutral species which possesses an unpaired electron, and which can be formed by splitting a molecule into to neutral fragments.Free radicals are generally very reactive due to the presence if an unpaired electron, and an incomplete valence shell.
Three examples of free radicals are sown below, oxygen free radical, chlorine free radical and bromine free radical
Due to the relatively high concentration of O2 in the atmosphere, the O radicals quickly combine to form ozone. This is because of the presence of the unpaired electron and their incomplete valence shell. As a result these O radicals are highly reactive and combine rapidly with O2 to form ozone. In the stratosphere, oxygen radical atoms are formed by the action of short wavelength UV radiation on O2 and O3. These O radicals will react with the excess of O2 to form O3, and then with O2 to reform O3
The main source of oxygen radical formation in the lower troposphere is due to the effect of sunlight breaking up ozone molecules and the emission of photochemical smog.
Oxygen radical formation in the stratosphere occurs due to the reaction that take place between oxygen molecule and UV light, this is further explained in the previous section, under the heading Ozone – good or bad?
- - - - --
The high reactivity of O atoms mean that their concentration in the stratosphere is consequently very low. Hypothetically, if there were high concentrations on O atoms they would readily recombine to form O2, and as a result there would be no available O atoms. Due to their short presence as single atoms, we cannot measure certain properties such as boiling and melting points.
- - - - --
The high reactivity of O atoms mean that their concentration in the stratosphere is consequently very low. Hypothetically, if there were high concentrations on O atoms they would readily recombine to form O2, and as a result there would be no available O atoms. Due to their short presence as single atoms, we cannot measure certain properties such as boiling and melting points.
| O (oxygen free radical) | O2 (oxygen gas) | O3 (ozone) | |
| Reactivity | Highly reactive | Moderately reactive | Highly reactive |
| Decomposed by high energy UV light | Decomposed by medium energy UV light | ||
| Structure | Each radical contains two unpaired valence shell electrons | Diatomic molecule – two oxygen atoms held together with a double covalent bond | Three oxygen atoms held together with one single coordinate covalent bond and one double covalent bond |
| Sing atom in a molecule | Molecule is linear shape | Molecular shape is bent |