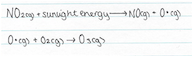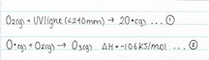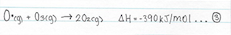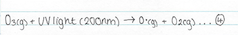The Structure and Composition of the Atmosphere
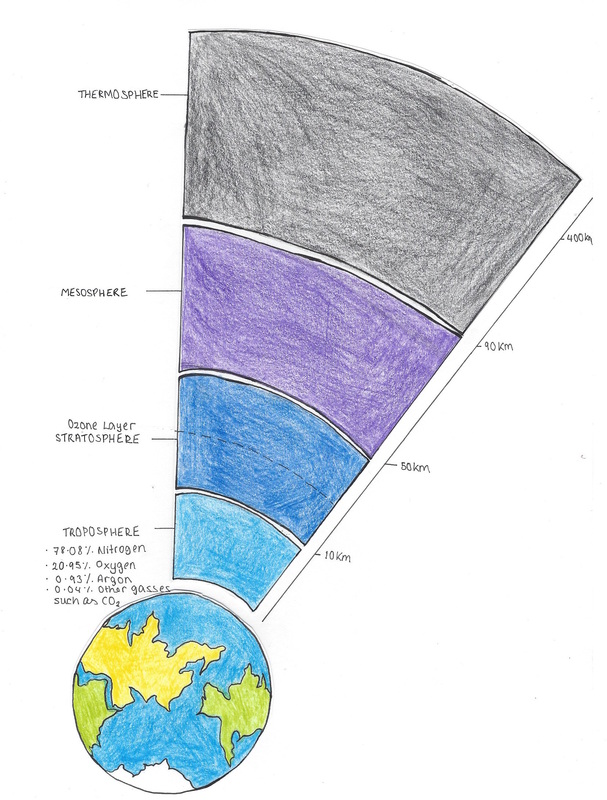
4.1 Present a quick sketch of the atmosphere labeling each layer (showing approximate thicknesses of each layer) and identifying where the “ozone layer” is (which atmospheric layer). For the troposphere note the four main gases and their percentage.
Atmospheric Pollutants
Identify the main pollutants found in the lower atmosphere and their sources
Describe ozone as a molecule able to act both as an upper atmosphere UV radiation shield and a lower atmosphere pollutant
Describe ozone as a molecule able to act both as an upper atmosphere UV radiation shield and a lower atmosphere pollutant
| Atmospheric Pollutant | Brief Summary | Sources of the pollutant | Effects of the pollutant on human health and the environment | |
| Artificial | Natural | |||
| Carbon monoxide (CO) | Carbon monoxide is a common dangerous and toxic gas; it is similar in size to dioxygen molecules. In the atmosphere, the carbon monoxide is oxidized to form carbon dioxide | Incomplete combustion of fuels | Decomposition of plants and animals | Carbon monoxide combines readily and rapidly with haemoglobin in the blood, due to its size relative to dioxygen molecules. The carbon monoxide takes the place of oxygen when binding to haemoglobin inhibiting the uptake of oxygen, causing a condition known as carbon monoxide poisoning. |
| Carbon monoxide reacts with other pollutants in the air to form potentially harmful ground level ozone. This occurs close to the site of emission. | ||||
| Carbon dioxide (CO2) | Carbon dioxide is a comparatively normal component of the atmosphere, although its concentrations have exponentially increased since the Industrial revolution. | Combustion of fuels in motor vehicles | Decomposition of plants and animals | Carbon dioxide is theorized to be a major cause of the increase in global warming, due to the greenhouse effect. |
| Carbon dioxide in its gas form is an asphyxiant, which cuts off the oxygen supply for breathing, especially in confined spaces. Exposure to high concentrations can cause death, unconsciousness, or convulsions. Exposure may damage a developing fetus. | ||||
| Nitrogen oxides (NOx) | Nitrogen oxides consist of nitric oxide, nitrogen dioxide and nitrous oxide. They are formed when nitrogen (N2) combines with oxygen (O2). Their lifespans in the atmosphere range from one to seven days for nitric oxide and nitrogen dioxide, to 170 years for nitrous oxide. | Motor vehicles | Soil Bacteria | As one of the components of smog, nitrogen dioxide is known to irritate the lungs and increase susceptibility to respiratory infections. |
| Nitric oxide has no colour, odor, or taste and is non-toxic. In the air it is rapidly oxidized to nitrogen dioxide. | Nitrogen oxides in the air can significantly contribute to a number of environmental effects such as acid rain and eutrophication in coastal waters | |||
| Sulfur dioxide (SO2) | Sulfur dioxide (SO2) is one of a group of highly reactive gasses known as oxides of sulfur. Sulfur dioxide dissolves easily in water to form sulfuric acid. | Electricity production | Bacteria | Sulfur dioxide is an irritant. It affects the nose, throat, and airways to cause coughing, wheezing, shortness of breath, or a tight feeling around the chest. |
| Sulfuric acid is a major component of acid rain. | ||||
| Lead | Lead is a naturally occurring heavy metal that is found in the Earth’s crust. Lead can be released into soil, air and water through soil erosion, volcanic eruptions, sea spray and bushfires. The natural concentration of lead in the air is less than 0.1 microgram per cubic meter. | Incinerators | Biomass burning | Once in the body, lead circulates in the blood. The amount of lead in a person's blood gives an indication of how much lead has recently been breathed in or swallowed. These measurements are called blood lead levels. Moderate-high lead levels in the blood act as neurotoxins interfere with nerve and brain functions and can cause infertility. |
| Particulate matter | Particulate matter, also known as particle pollution, is a complex mixture of extremely small particles and liquid droplets. Particle pollution is made up of a number of components, including acids (such as nitrates and sulfates), organic chemicals, metals, and soil or dust particles. | Combustion of fossil fuels | Biomass burning | The microscopic solids or liquid droplets that are so small that they can get deep into the lungs and cause decreased lung functioning including asthma, coughing and irritation of the airways. |
| (particles) | Deposition of fine particulate matter leads to acid deposit issues in the environment. | |||
| Volatile Organic Compounds (VOC) | Volatile organic compounds (VOCs) are emitted as gases from certain solids or liquids. VOCs include a variety of chemicals. VOCs are emitted by a wide array of products numbering in the thousands. | Un-burnt fuel | Emitted by vegetation | VOC are eyes and ENT irritants, causing headaches, vertigo, visual disorders and memory impairment |
| VOC’s have been found to be to be a major contributing factor in the production of ozone, as in the air react with oxides of nitrogen in the presence of sunlight to form ozone, which inhibit the troposphere. | ||||
Ozone - Good or Bad?
4.2 Identify the main pollutants found in the lower atmosphere and their sources
4.3 Describe ozone as a molecule able to act both as an upper atmosphere UV radiation shield and a lower atmosphere pollutant
Ozone is naturally present in the atmosphere and is concentrated around 2ppm (parts per million) by volume. Near ground level; its concentration of clean air is less than 0.02ppm (parts per million) by volume of ozone in the stratosphere the presence of ozone ranges from 2-8ppm and then back to 2ppm as the altitude changes from around 15km to 30km. The concentration of ozone in the atmosphere sitting being less than 2ppm is a small quantity and concentration in comparison to other major gasses, such as oxygen (2.0 x 108 ppm) and Nitrogen (8.8 x 108 ppm). All the ozone in the atmosphere would form a thin layer approximately 3mm thick at 0°C and 1 atmospheric pressure.
4.3 Describe ozone as a molecule able to act both as an upper atmosphere UV radiation shield and a lower atmosphere pollutant
Ozone is naturally present in the atmosphere and is concentrated around 2ppm (parts per million) by volume. Near ground level; its concentration of clean air is less than 0.02ppm (parts per million) by volume of ozone in the stratosphere the presence of ozone ranges from 2-8ppm and then back to 2ppm as the altitude changes from around 15km to 30km. The concentration of ozone in the atmosphere sitting being less than 2ppm is a small quantity and concentration in comparison to other major gasses, such as oxygen (2.0 x 108 ppm) and Nitrogen (8.8 x 108 ppm). All the ozone in the atmosphere would form a thin layer approximately 3mm thick at 0°C and 1 atmospheric pressure.
| Per cent of atmosphere by mass | Total mass (g) | Number of moles | Volume at STP | Molar mass | Parts per million | |
| Oxygen | 23.1 | 1.18 x 1021 | 3.68 x 1019 | 8.24 x 1020 | 32 | 0.23 x 106 |
| Nitrogen | 76.9 | 3.92 x 1021 | 1.4 x 1020 | 3.14 x 1021 | 28 | 0.78 x 106 |
- - - - -
The troposphere is the region of the atmosphere closest to the earth, in which temperature generally decreases as altitude increases: it extends from sea level to an altitude of about 15km. In the troposphere the concentration of ozone in clean air is only 0.02ppm, which is approximately 10% of all ozone. 10% of the total ozone is found is present the troposphere where is contributes to pollution and affects health. The main source of ozone in the troposphere is due to the effect of sunlight in photochemical smog. Smog, or photochemical smog is a type of air pollution that is produce when sunlight acts upon motorcar exhaust gases to form ozone and other harmful substances.
- - - - -
Ozone forms in the lower atmosphere (the troposphere) where sunlight concentration is abundant, especially in the summer seasons, and when the nitrogen dioxide (NO2) concentrations are well above clean air levels. The formation of ozone occurs when there is no breeze, or small wind, to disperse the pollutants amongst the air. The sunlight breaks of an oxygen atom from the NO2 molecule, forming oxygen free radical. This free radical of oxygen is highly unstable and combines with an oxygen molecule (O2) to form ozone.
The rate at which this reaction can occur is dependent upon the concentration of the reactants. As a result, if the concentration of NO2(g) is low, and the concentration of NO(g) is high, the rate of the reaction between the NO(g) and O3(g) reaction is much greater than the rate of the NO2(g) and light reaction, and consequently, the newly formed ozone is destroyed at a rate which matches that of its formation, and as a result keeping its concentration below harmful levels.
- - - - -
Ozone is a pollutant in the lower levels of the atmosphere. Tropospheric ozone, as previously discussed, represents approximately 10% of the quantity of the overall ozone in the atmosphere. We know that it is generated by nitric oxides (NO2 and NO3). Ground level ozone is very poisonous at concentrations above 20ppmas it readily oxidizes with organic tissues, disrupting the normal biochemical reactions that occur in the body. Ozone is an irritant to both the eyes and lungs, and is toxic to plants (including crops). The strength of ozone as an oxidizing agent is much stronger of that of oxygen gas (O2), this strength is heightened in environments that are acidic (pH<7). Ozone will readily attack rubbers and plastics. There is a saying that goes “Ozone is good up high, but bad nearby.”
- - - - -
In order to reduce ground level ozone, we must minimize photochemical smog formation. To minimize the formation of photochemical smog, the government has implemented maximum permissible exhaust emissions for both oxides of nitrogen and hydrocarbons, alongside carbon monoxide, which is poisonous without further reaction. In Australia, the maximum allowable exhaust emission for any size passenger car has lead to reductions of 90%. Since 1986, new cars in Australia have been fitted with catalytic exhausts. The ones fitted at the moment convert NO into N2 and hydrocarbons into and carbon monoxide into carbon dioxide.
- - - - -
The ozone formation-destruction equilibrium is the balance between the formation and destruction of oxygen, in the following reactions, which result in a ‘steady state’ concentration of ozone in the stratosphere (a steady state is a situation in which all state variables are constant in spite of ongoing processes that strive to change them).
- - - - -
Ozone is a pollutant in the lower levels of the atmosphere. Tropospheric ozone, as previously discussed, represents approximately 10% of the quantity of the overall ozone in the atmosphere. We know that it is generated by nitric oxides (NO2 and NO3). Ground level ozone is very poisonous at concentrations above 20ppmas it readily oxidizes with organic tissues, disrupting the normal biochemical reactions that occur in the body. Ozone is an irritant to both the eyes and lungs, and is toxic to plants (including crops). The strength of ozone as an oxidizing agent is much stronger of that of oxygen gas (O2), this strength is heightened in environments that are acidic (pH<7). Ozone will readily attack rubbers and plastics. There is a saying that goes “Ozone is good up high, but bad nearby.”
- - - - -
In order to reduce ground level ozone, we must minimize photochemical smog formation. To minimize the formation of photochemical smog, the government has implemented maximum permissible exhaust emissions for both oxides of nitrogen and hydrocarbons, alongside carbon monoxide, which is poisonous without further reaction. In Australia, the maximum allowable exhaust emission for any size passenger car has lead to reductions of 90%. Since 1986, new cars in Australia have been fitted with catalytic exhausts. The ones fitted at the moment convert NO into N2 and hydrocarbons into and carbon monoxide into carbon dioxide.
- - - - -
The ozone formation-destruction equilibrium is the balance between the formation and destruction of oxygen, in the following reactions, which result in a ‘steady state’ concentration of ozone in the stratosphere (a steady state is a situation in which all state variables are constant in spite of ongoing processes that strive to change them).
Oxygen atoms can also react with ozone
As a result the ozone absorbs longer wavelengths of UV, which decompose the ozone back into dioxygen gas.
The formation-destruction equilibrium, and the above four reactions, explain why the temperature in the stratospheres is greater than that in the tropopause, despite its distance from the earth. The reactions labeled 1 and 4 absorb UV radiation and reactions 2 and 3 release the UV energy, and as a result, warm up the air.
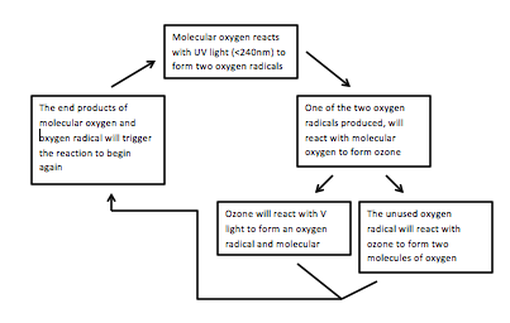
The ability of ozone formation-destruction equilibrium to absorb UV in the stratosphere is due to the breaking of the covalent bonds in dioxygen via UV radiation (less than 240nm) into two oxygen radicals. The oxygen radical will consequently react with the dioxygen molecule to form ozone. This ozone molecule will then again react with UV and produce another oxygen radical and oxygen molecule. The oxygen radical will then react with another ozone molecule to form two dioxygen molecules, which triggers the process to start again. The breaking of the bonds in both the dioxygen molecule and the ozone molecule, absorb UV, preventing harmful UV rays from reaching the surface of the earth. This is best represented in the flow chart below
The ability of ozone formation-destruction equilibrium to absorb UV in the stratosphere is due to the breaking of the covalent bonds in dioxygen via UV radiation (less than 240nm) into two oxygen radicals. The oxygen radical will consequently react with the dioxygen molecule to form ozone. This ozone molecule will then again react with UV and produce another oxygen radical and oxygen molecule. The oxygen radical will then react with another ozone molecule to form two dioxygen molecules, which triggers the process to start again. The breaking of the bonds in both the dioxygen molecule and the ozone molecule, absorb UV, preventing harmful UV rays from reaching the surface of the earth. This is best represented in the flow chart below
- - - - -
Stratospheric ozone is helpful to biological processes, which occur on the earth’s surface. Stratospheric ozone absorbs harmful UV rays that are less than 240nm. These wavelengths are very likely to cause mutations, such as sunburn, which can lead to diseases such as skin cancer, which can corrupt biological DNA. Other examples of biological mutations which occur from overexposure to UV radiation includes eye cataracts, the lowering of the immune system (increasing the risk of infection). Overexposure to UV radiation can reduce plant growth for some species due to the UV interference with their biological mechanisms such as their mechanism for Photosynthesis. Rice is a particular example of a species that is particularly vulnerable to damage by UV radiation.
- - - - -
The wavelengths of UV lights vary. The shorter the wavelength’s the more dangerous and damaging the results from the radiation is on biological processes. Stratospheric ozone absorbs these damaging UV wavelengths, preventing them from reaching the surface of the earth.
Stratospheric ozone is helpful to biological processes, which occur on the earth’s surface. Stratospheric ozone absorbs harmful UV rays that are less than 240nm. These wavelengths are very likely to cause mutations, such as sunburn, which can lead to diseases such as skin cancer, which can corrupt biological DNA. Other examples of biological mutations which occur from overexposure to UV radiation includes eye cataracts, the lowering of the immune system (increasing the risk of infection). Overexposure to UV radiation can reduce plant growth for some species due to the UV interference with their biological mechanisms such as their mechanism for Photosynthesis. Rice is a particular example of a species that is particularly vulnerable to damage by UV radiation.
- - - - -
The wavelengths of UV lights vary. The shorter the wavelength’s the more dangerous and damaging the results from the radiation is on biological processes. Stratospheric ozone absorbs these damaging UV wavelengths, preventing them from reaching the surface of the earth.