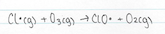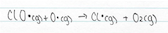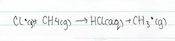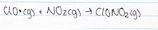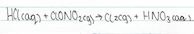Origin's of Chlorofluorocarbons (CFC's)
4.10 Identify the origins of chlorofluorocarbons (CFC's) and halons in the atmosphere
A haloalkane is a compound in which one or more H atom/s of an alkane has been replacd by a halogen atom. A halogen atom is any element which belongs to the halogen group on the periodic table.
CFC’s or chlorofluorocarbon’s are compounds containing carbon, chlorine and fluorine only – as a result, the contain no hydrogen.
A halon is a group of chemical compounds consisting of alkanes with linked halogens. In particular, bromine-containing haloalkanes.
A haloalkane is a compound in which one or more H atom/s of an alkane has been replacd by a halogen atom. A halogen atom is any element which belongs to the halogen group on the periodic table.
CFC’s or chlorofluorocarbon’s are compounds containing carbon, chlorine and fluorine only – as a result, the contain no hydrogen.
A halon is a group of chemical compounds consisting of alkanes with linked halogens. In particular, bromine-containing haloalkanes.
| Common name | CFC-113 |
| Systematic name | 1,1,2-trichloro-1,2,2-trifluroethane |
| Use | Cleaning electronic circuit boards |
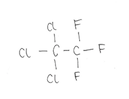
CFC-113 consists of two carbon atoms, three chlorine atoms and three fluorine atoms and looks like:
CFC’s were originally introduced under the tradename name ferons, in the 1930’s to replace toxic ammonia as a refrigerant gas. CFC’s hold several advantages over ammonia, as they are unreactive and non-toxic molecules, and readily liquefied upon compression. Their boiling point and high dependence on pressure made them idea working fluids. Above ammonia, CFC’s are odorless, non-flammable, non-toxic and inert, which made them exceedingly more desirable than the ammonia which they replaced. After World War 1, applications of CFC’s were becoming increasingly important, they became widely used in aerosol spray cans as the propelling ages, then later they became the foaming/blowing agents to expand many plastics, such as polystyrene foam and as solvents in the cleaning of electrical circuits.
A Halon is an unreactive gaseous compounds of carbon with bromine and other halogens initially, they were developed for and used in fire extinguishers, particularly in large computer systems and on airplanes. However the small yellow ones which were commonly used in cars and boats were banned in 1994.
As CFC’s were odorless, non-flammable, non-toxic and very inert, they were a very attractive molecule when they were first released in the 1930’s. As CFC’s were used in aerosol spray cans, cleaning of electrical circuit boards and as foaming agents, their release was directly into the atmosphere. This release into the atmosphere was considered of no concern because FC’s were non-toxic and inert.
However, it was the inertness combined with their insolubility in water (rain), which caused them to become a problem. CFC’s were not destroyed by sunlight energy or oxygen (as most organic compounds in the atmosphere are), they spread out in uniform around the world, until their concentration in the stratosphere gradually began to cause the hole in the ozone layer.
A Halon is an unreactive gaseous compounds of carbon with bromine and other halogens initially, they were developed for and used in fire extinguishers, particularly in large computer systems and on airplanes. However the small yellow ones which were commonly used in cars and boats were banned in 1994.
As CFC’s were odorless, non-flammable, non-toxic and very inert, they were a very attractive molecule when they were first released in the 1930’s. As CFC’s were used in aerosol spray cans, cleaning of electrical circuit boards and as foaming agents, their release was directly into the atmosphere. This release into the atmosphere was considered of no concern because FC’s were non-toxic and inert.
However, it was the inertness combined with their insolubility in water (rain), which caused them to become a problem. CFC’s were not destroyed by sunlight energy or oxygen (as most organic compounds in the atmosphere are), they spread out in uniform around the world, until their concentration in the stratosphere gradually began to cause the hole in the ozone layer.
Problems Caused by CFC's
4,8 Discuss the problems
associated with the use of CFCs and assess the effectiveness of steps taken to
alleviate these problems
The ozone hole, or stratospheric ozone depletion was first discovered in 976 when the British Antarctic Survey at Halley Bay notice that there was a 10% drop in the levels of stratospheric ozone over the Antarctic in the southern spring. The ‘ozone hole’ is the thinning of the ozone layer to the extent where the CFC’s in the atmosphere are destroying and thinning the layer enough to cause essentially, a hole (located over Antarctica).
Globally, CFC’s cause a 3-8% decrease in the amount of stratospheric ozone per annum, so stratospheric ozone depletion is not limited to the Antarctica, however the Antarctic sees more decrease in stratospheric ozone (50-90% decrease over the spring).
Ozone depletion refers to the 3-8% decrease in the amount of ozone in the stratosphere caused by CFC’s, this occurs on a year round basis, however, the ozone hole is of much more concern as it is the hole caused by ozone depletion, resulting in a 50-90% decrase of stratospheric ozone during the Antarctic spring.
- - - - -
The ozone hole, or stratospheric ozone depletion was first discovered in 976 when the British Antarctic Survey at Halley Bay notice that there was a 10% drop in the levels of stratospheric ozone over the Antarctic in the southern spring. The ‘ozone hole’ is the thinning of the ozone layer to the extent where the CFC’s in the atmosphere are destroying and thinning the layer enough to cause essentially, a hole (located over Antarctica).
Globally, CFC’s cause a 3-8% decrease in the amount of stratospheric ozone per annum, so stratospheric ozone depletion is not limited to the Antarctica, however the Antarctic sees more decrease in stratospheric ozone (50-90% decrease over the spring).
Ozone depletion refers to the 3-8% decrease in the amount of ozone in the stratosphere caused by CFC’s, this occurs on a year round basis, however, the ozone hole is of much more concern as it is the hole caused by ozone depletion, resulting in a 50-90% decrase of stratospheric ozone during the Antarctic spring.
- - - - -
| UVa | UVb | |
| Wavelength | 400-320nm | 320-280nm |
| Good or bad? | Beneficial | Harmful |
| Why? | Facilitates photosynthesis | Cuases mutations such as skin cancer and eye cataracts |
| Helps to form Vitamin D in humans | Decreases immune response | |
| Damages plants | ||
| Severe sun burn |
Solar ultraviolet radiation is frequently divided into three categories: UVa, UVb and UVc, increasing harmful wavelengths in the same order. UVa occurs within the wavelengths of UV light 400-320nm and is generally considered to be beneficial to life on earth, as it facilitates photosynthesis and aids to form Vitamin D in humans. UVb occurs in the wavelengths 320 to 280nm and is considered harmful to humans as it causes biological mutations such as skin cancer, eye cataracts and decreases immune response, so there is an increase in disease, alongside being damaging to humans, it is also damaging to plants.
Ozone prevents harmful UVb rays from reaching earths surface as it absorbs UVb rays as they try to pass through it. Ozone depletion poses a problem as there is little ozone for the UVb rays to react with, thus they are not absorbed and can each the earths surface.
Stratospheric ozone filters out most of the sun's potentially harmful shortwave ultraviolet (UV) radiation. If this ozone becomes depleted, then more UV rays will reach the earth. Exposure to higher amounts of UV radiation could have serious impacts on human beings, animals and plants, such as the following:
Harm to human health:
· More skin cancers, sunburns and premature aging of the skin.
· More cataracts, blindness and other eye diseases: UV radiation can damage several parts of the eye, including the lens, cornea, retina and conjunctiva.
· Cataracts (a clouding of the lens) are the major cause of blindness in the world. A sustained 10% thinning of the ozone layer is expected to result in almost two million new cases of cataracts per year, globally (Environment Canada, 1993).
· Weakening of the human immune system (immunosuppression). Early findings suggest that too much UV radiation can suppress the human immune system, which may play a role in the development of skin cancer.
Impacts on Plants
· Only a few commercially important trees have been tested for UV (UV-B) sensitivity, but early results suggest that plant growth, especially in seedlings, is harmed by more intense UV radiation.
Damage to marine life:
· In particular, plankton (tiny organisms in the surface layer of oceans) are threatened by increased UV radiation. Plankton are the first vital step in aquatic food chains.
· Decreases in plankton could disrupt the fresh and saltwater food chains, and lead to a species shift in Canadian waters.
· Loss of biodiversity in our oceans, rivers and lakes could reduce fish yields for commercial and sport fisheries.
Synthetic materials:
· Wood, plastic, rubber, fabrics and many construction materials are degraded by UV radiation.
· The economic impact of replacing and/or protecting materials could be significant.
- - - - -
When in the stratosphere, CFC’s come into contact with short wavelength UV radiation (before ozone has filtered the UV out) and this short wavelength UV breaks a chlorine atom of a CFC molecule
The chlorine atom that is formed when CFC’s react with UV light, abstracts as an oxygen atom to form an ozone molecule, forming a ClO free radical
Like all free radicals, the ClO, which has been formed, is very reactive. Due to the continual formation and destruction of the ozone in the stratosphere, there are free oxygen atoms present (along side O2 and O3 molecules). ClO reacts with a free oxygen atom to form a molecule of oxygen and O2 and this process regenerates the chlorine atom
The following result is the sum of the two above equations, which is the fact an ozone molecule and an oxygen atom have been converted into two oxygen molecules and the relative chlorine atom has still not been used up. This single chlorine atom acts as a catalyst for ozone destruction and is able to repeat this process indefinitely. This continual repetition is the process of Chain Reactions. Meaning that chlorine free radical has the ability to destroy all ozone in the stratosphere.
- - - - -
The chlorine free radical chain reaction can be naturally halted in to ways, the first being the reaction between a chlorine free radical atom an a methane molecule. Small quantities of methane are present in the stratosphere, due to the slow process of diffusion up from the troposphere, this reaction will form hydrogen chloride, eliminating the chlorine free radical and bringing the chain reaction to a halt.
- - - - -
The chlorine free radical chain reaction can be naturally halted in to ways, the first being the reaction between a chlorine free radical atom an a methane molecule. Small quantities of methane are present in the stratosphere, due to the slow process of diffusion up from the troposphere, this reaction will form hydrogen chloride, eliminating the chlorine free radical and bringing the chain reaction to a halt.
Another way in which the chlorine chain reaction can be halted involves the ClO species. Small amount of nitrogen dioxide are present in the stratosphere. Chlorine nitrate is formed when a ClO radical hits a nitrogen dioxide molecule. The occurrence of this reaction essentially removes the ClO from the reaction and brings the destruction of ozone to a halt, buy preventing further chain reactions to occur
- - - - -
Ozone depletion over Antarctica in winter is for two reasons, as winter in Antarctica is a period of continuous darkness and the stratosphere is extremely cold. Under the extremely cold conditions certain solid particles forms that are unable to catalyze a reaction between chlorine nitrate and hydrogen chloride.
Ozone depletion over Antarctica in winter is for two reasons, as winter in Antarctica is a period of continuous darkness and the stratosphere is extremely cold. Under the extremely cold conditions certain solid particles forms that are unable to catalyze a reaction between chlorine nitrate and hydrogen chloride.
The conversion of hydrogen chloride and chlorine nitrate to molecular chlorine (Cl2) has no affect upon ozone concentrations during winter. As molecular chlorine cannot undergo further reactions (due to the extreme cold and darkness), it is unable to produce two chlorine free radicals, which will react further to destroy ozone.
In the spring when the sun comes up, the level of ozone depletion switches. Sunlight is available and splits the chlorine molecule into two chlorine atoms, each of which can destroy the ozone,
In the spring when the sun comes up, the level of ozone depletion switches. Sunlight is available and splits the chlorine molecule into two chlorine atoms, each of which can destroy the ozone,
The concentration of ozone above Antarctica is greatly reduced; consequently the ozone hole is formed.
In the summer the polar vortex breakes up so thre is mixing of Antarctic air with lower-latitude air. This returns the ozone concentration back to normal. The break up of the polar vortex has occasionally influenced countries in the southern hemisphere, as it can bring ozone depleted stratospheric air over these southern countries and states (particularly Tasmania and Victoria) in the early summer.
In the summer the polar vortex breakes up so thre is mixing of Antarctic air with lower-latitude air. This returns the ozone concentration back to normal. The break up of the polar vortex has occasionally influenced countries in the southern hemisphere, as it can bring ozone depleted stratospheric air over these southern countries and states (particularly Tasmania and Victoria) in the early summer.
Reactions in the Atmosphere
 Karin L Gleeson, (2008), A Photo of the Dobson Spectrometer [ONLINE]. Available at: http://www.ozonelayer.noaa.gov/action/Dobson_Bldr_Apr01.jpg [Accessed 04 March 15].
Karin L Gleeson, (2008), A Photo of the Dobson Spectrometer [ONLINE]. Available at: http://www.ozonelayer.noaa.gov/action/Dobson_Bldr_Apr01.jpg [Accessed 04 March 15].
4.12 Present information from secondary sources to write the equations to show the reactions involving CFC's and ozone to demonstrate the removal of ozone from the atmosphere
In Australia, the Bureau of Meteorology monitors the stratospheric ozone through the use of ground-based instruments, satellites, balloons and aircraft.
Dobson Spectrometers; or UV spectrometers to monitor stratospheric ozone throughout five main sights: Perth, Darwin, Melbourne, Brisbane and Macquarie Island. These instruments are pointed vertically upwards through the atmosphere and measure the quantity of light intensity received at a wavelength at which ozone absorbs, and then a wavelength either sides of this at which ozone does not absorb. A comparison of the varieties of intensities collected gives a measure of the total ozone in the atmosphere per unit area of the Earths surface.
Umkehrs; which are a sequence of special ozone measurements made from sunrise to sunset using a spectrometer. These measurements provide a crude atmospheric ozone profile at 9 levels (0-49km). Unfortunately, for these measurement to occur, they require clear skies.
The Ozone Sonde; are balloon-borne instruments that continuously estimate ozone concentrations as they ascend into the atmosphere. A profile of ozone is obtained up to the burst point of the balloon, in excess of 30km
- - - - -
In addition to the ground based equipment the Bureau also conducts monitoring using satellite data, which is performed by NASA. NASA routinely retrieves analysis and archives total ozone data from the Tiros Operational Vertical Scanner (TOVS). This data comes in from the National Oceanic and Atmospheric Administrative (NOAA) team's polar orbiting satellites. Which have provided data from 19 October, 1978 to the present. This form of data is received routinely via Global Telecommunications Systems, they are analysed and converted into images. The ozone analysis are used as input for the Bureau’s UV analysis and forecasting scheme.
There is other, more accurate satellite data available from the Total Ozone mapping Spectrometer (TOMS) and Solar Backscatter UV (SBUV) however these are not available in ‘real time’ and are therefore no longer used used. The Ozone Mapping Profiler Suite (OMPS) is most frequently used, as it is an advances suite of two hyper spectral instruments. OMPS extends the 25 year total-ozone and ozone-profile records, which are used by ozone-assessment researchers and policy makers to track the health of the ozone layer. OMPS has improved vertical resolution data and allows for better testing and monitoring of the chemistry involved in ozone destruction near the troposphere.
In Australia, the Bureau of Meteorology monitors the stratospheric ozone through the use of ground-based instruments, satellites, balloons and aircraft.
Dobson Spectrometers; or UV spectrometers to monitor stratospheric ozone throughout five main sights: Perth, Darwin, Melbourne, Brisbane and Macquarie Island. These instruments are pointed vertically upwards through the atmosphere and measure the quantity of light intensity received at a wavelength at which ozone absorbs, and then a wavelength either sides of this at which ozone does not absorb. A comparison of the varieties of intensities collected gives a measure of the total ozone in the atmosphere per unit area of the Earths surface.
Umkehrs; which are a sequence of special ozone measurements made from sunrise to sunset using a spectrometer. These measurements provide a crude atmospheric ozone profile at 9 levels (0-49km). Unfortunately, for these measurement to occur, they require clear skies.
The Ozone Sonde; are balloon-borne instruments that continuously estimate ozone concentrations as they ascend into the atmosphere. A profile of ozone is obtained up to the burst point of the balloon, in excess of 30km
- - - - -
In addition to the ground based equipment the Bureau also conducts monitoring using satellite data, which is performed by NASA. NASA routinely retrieves analysis and archives total ozone data from the Tiros Operational Vertical Scanner (TOVS). This data comes in from the National Oceanic and Atmospheric Administrative (NOAA) team's polar orbiting satellites. Which have provided data from 19 October, 1978 to the present. This form of data is received routinely via Global Telecommunications Systems, they are analysed and converted into images. The ozone analysis are used as input for the Bureau’s UV analysis and forecasting scheme.
There is other, more accurate satellite data available from the Total Ozone mapping Spectrometer (TOMS) and Solar Backscatter UV (SBUV) however these are not available in ‘real time’ and are therefore no longer used used. The Ozone Mapping Profiler Suite (OMPS) is most frequently used, as it is an advances suite of two hyper spectral instruments. OMPS extends the 25 year total-ozone and ozone-profile records, which are used by ozone-assessment researchers and policy makers to track the health of the ozone layer. OMPS has improved vertical resolution data and allows for better testing and monitoring of the chemistry involved in ozone destruction near the troposphere.
| Technique | Advantages | Disadvantages |
| Dobson Spectrometers | By comparing the UV light intensity at wavelengths that are strongly absorbed and weakly absorbed by ozone, the ozone content of the atmosphere is accurately determined | Inability to make accurate measurements at high total ozone amounts |
| Umkehrs | Higher spatial resolution | Runs on a seasonal cycle |
| Ozone Sonde | Light weight and relatively cheep | Cannot measure high altitudes |
| TOMS | Accurate satellite data | Does not relay real time information and are therefore useless |
| OMPS | Two hyper spectral instruments resulting in highly accurate | None have been found as of yet |
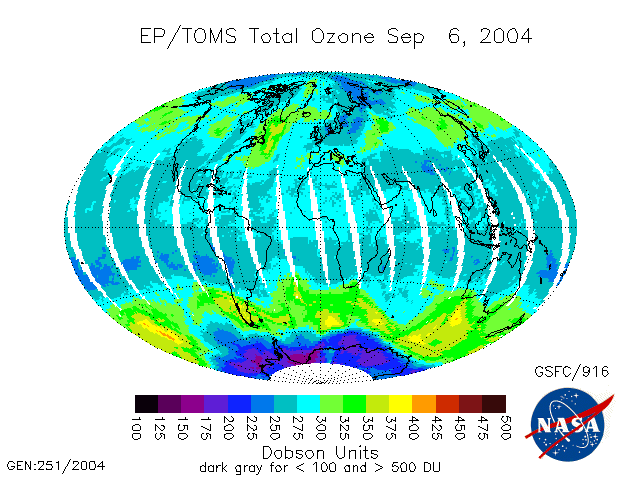
The total ozone per unit area above a location is typically measured in Dobson Units. A Dobson unit is the amount of ozone that corresponds to a 0.01mm thickness of pure ozone at atmospheric pressure. The normal amount of ozone is 300DU.
The Montreal Protocol and Alternatives to CFC’s
4.14 Present informations from secondary sources to identify alternative chemicals used to replace CFC's and evaluate the effectiveness of their use as a replacement for CFC's
A HCFC is a hydrochloroflurocarbon, these compounds contain only the elements carbon, hydrogen, fluorine and chlorine
The presence of hydrogen in HCFC’s makes them more reactive, which allows them to be destroyed in the troposphere, preventing them from reaching the stratosphere and therefore they cannot reach the ozone layer. HCFC
There are four main reasons why HCFC’s are considered to be a temporary replacement for CFC’s, the four reasons are
1. The suitability of the properties of replacement chemicals as to their application
HCFC’s are toxic, flammable, corrosive and explosive than the CFC’s, which they replace, however they are appropriate coolants
2. The ozone depletion potential of the replacement chemicals
The reactions which destroy HCFC’s are quite slow, so a substantial proportion if intact molecule will reach the stratosphere where
they have ODP
The HCFC molecules contain chlorine, and thus, C-Cl bonds. In the stratosphere radiation can break these bonds leading to Cl free
radical emission, starting a chain reaction which will slowly destroy the ozone layer, this is less, but still destructive which will lead to
them being phased out
However, HCFC’s will only 1 to 10% as much ozone and the same amount of CFC,
3. Global warming potential or other hazards of the replacement chemicals
GWP of 1000 times that of CO2
HCFC’s are green house gasses and will contribute significantly to global warming
4. The cost of production of alternatives or cost of making changes to existing equipment
HCFC’s require massive modifications in equipment, which is expensive, however the only alternatives are inefficient cooling agents
The process of phasing out the CFC’s and introducing these alternatives is costly process and hence many developing countries cannot
afford this cost
- - - - -
HFC’s are hydroflurocarbons meaning that they are compounds which contain only elements of hydrogen, fluorine and carbon.
HFC’s contain C-H bonds and as a result undergo some decomposition in the stratosphere, meaning they have zero effect on the ozone layer. Unlike HCFC’s, HFC’s contain no chlorine at all, so they have a zero ozone depletion potential, even if they do reach the stratosphere.
The global warming potential of HFC’s is a large disadvantage relating to the compound. HFC’s are essentially greenhouse gasses and therefore contribute significantly to global warming; they absorb heat radiation from the earth’s surface at a much greater rate than carbon dioxide (the standard), some up to 10,000 times. HFC-134a offers a nonflammable alternative to CFC’s, but unfortunately this is three times the cost, despite this HFC possessing suitable properties, however its GWP has been found to be 1,200 times more damaging that carbon dioxide.
Haloalkanes with some hydrogen atoms are now used. Initially HCFC's were used. These are less stable that CFC's due to the presents of a C-H bond, so these compounds are more likely to break down in the troposphere. However, it was found that some were still reaching the stratosphere and releasing chlorine radicals there. HFC's are now used.
There are a variety of HCFC’s and HFC’s, which are able to replace all CFC’s, these include:
Substitute CFC-113 (C2Cl3F3) used as an aerosol solvent
· C5 – C20 petroleum hydrocarbons
· Most HCFC’s, such asHCFC-141b
· HFC’s, such as HFC-25fa
Substitute for HFC-11 (CCl3F) used as and aerosol propellant
· Light hydrocarbons (C3 – C6)
· HFC’s, such as HFC-152a and HFC-134a
Substitute for CFC-11 (CCl3F) used in commercial refrigerators and insulating foam
· HCFC-123
· HCFC-141b
Substitute for CFC-12 (CCl2F2)
· HCFC-22
· HFC-13a
· A three part blend of the above two and HFC-142b
- - - - -
With total removal of CFC’s from the atmosphere stratospheric ozone will recover, as the damage done to it is revisable. However, even if all nations remove all traces of CFC’s, it will still take 50-100 years for complete ozone recovery to occur. This is because there is such a high concentration of CFC’s in the troposphere, and will take a long time to diffuse up into the stratosphere and be destroyed.
An criteria for judging the effectiveness of HFC’s and HCFC’s goes as follows:
A HCFC is a hydrochloroflurocarbon, these compounds contain only the elements carbon, hydrogen, fluorine and chlorine
The presence of hydrogen in HCFC’s makes them more reactive, which allows them to be destroyed in the troposphere, preventing them from reaching the stratosphere and therefore they cannot reach the ozone layer. HCFC
There are four main reasons why HCFC’s are considered to be a temporary replacement for CFC’s, the four reasons are
1. The suitability of the properties of replacement chemicals as to their application
HCFC’s are toxic, flammable, corrosive and explosive than the CFC’s, which they replace, however they are appropriate coolants
2. The ozone depletion potential of the replacement chemicals
The reactions which destroy HCFC’s are quite slow, so a substantial proportion if intact molecule will reach the stratosphere where
they have ODP
The HCFC molecules contain chlorine, and thus, C-Cl bonds. In the stratosphere radiation can break these bonds leading to Cl free
radical emission, starting a chain reaction which will slowly destroy the ozone layer, this is less, but still destructive which will lead to
them being phased out
However, HCFC’s will only 1 to 10% as much ozone and the same amount of CFC,
3. Global warming potential or other hazards of the replacement chemicals
GWP of 1000 times that of CO2
HCFC’s are green house gasses and will contribute significantly to global warming
4. The cost of production of alternatives or cost of making changes to existing equipment
HCFC’s require massive modifications in equipment, which is expensive, however the only alternatives are inefficient cooling agents
The process of phasing out the CFC’s and introducing these alternatives is costly process and hence many developing countries cannot
afford this cost
- - - - -
HFC’s are hydroflurocarbons meaning that they are compounds which contain only elements of hydrogen, fluorine and carbon.
HFC’s contain C-H bonds and as a result undergo some decomposition in the stratosphere, meaning they have zero effect on the ozone layer. Unlike HCFC’s, HFC’s contain no chlorine at all, so they have a zero ozone depletion potential, even if they do reach the stratosphere.
The global warming potential of HFC’s is a large disadvantage relating to the compound. HFC’s are essentially greenhouse gasses and therefore contribute significantly to global warming; they absorb heat radiation from the earth’s surface at a much greater rate than carbon dioxide (the standard), some up to 10,000 times. HFC-134a offers a nonflammable alternative to CFC’s, but unfortunately this is three times the cost, despite this HFC possessing suitable properties, however its GWP has been found to be 1,200 times more damaging that carbon dioxide.
Haloalkanes with some hydrogen atoms are now used. Initially HCFC's were used. These are less stable that CFC's due to the presents of a C-H bond, so these compounds are more likely to break down in the troposphere. However, it was found that some were still reaching the stratosphere and releasing chlorine radicals there. HFC's are now used.
There are a variety of HCFC’s and HFC’s, which are able to replace all CFC’s, these include:
Substitute CFC-113 (C2Cl3F3) used as an aerosol solvent
· C5 – C20 petroleum hydrocarbons
· Most HCFC’s, such asHCFC-141b
· HFC’s, such as HFC-25fa
Substitute for HFC-11 (CCl3F) used as and aerosol propellant
· Light hydrocarbons (C3 – C6)
· HFC’s, such as HFC-152a and HFC-134a
Substitute for CFC-11 (CCl3F) used in commercial refrigerators and insulating foam
· HCFC-123
· HCFC-141b
Substitute for CFC-12 (CCl2F2)
· HCFC-22
· HFC-13a
· A three part blend of the above two and HFC-142b
- - - - -
With total removal of CFC’s from the atmosphere stratospheric ozone will recover, as the damage done to it is revisable. However, even if all nations remove all traces of CFC’s, it will still take 50-100 years for complete ozone recovery to occur. This is because there is such a high concentration of CFC’s in the troposphere, and will take a long time to diffuse up into the stratosphere and be destroyed.
An criteria for judging the effectiveness of HFC’s and HCFC’s goes as follows:
| HFC’s | HCFC’s | |
| Ozone depletion potential | Ozone depletion potential is zero as they contain no chlorine | Deplete the ozone by 10% compared to HFC’s, however they will be banned by 2030 |
| Global warming potential | Greenhouse gasses, however HFC’s have more impact than HCFC’s | Greenhouse gas |
| Cost of production | More expensive than CFC’s | |
| Effectiveness of substitute | Zero ODP, however not as affective compared to CFC’s | Still causes damage to the ozone, however are a more affective substitute for CFC’s than HFC’s |
HFC’s and HCFC’s have been a very effective replacement chemical for CFC's however they are no a perfect solution . These compounds are less stable than CFC's due to their CH bond, and so are broken don in the troposphere. They don't reach the stratosphere and therefore do not deplete ozone. However, as refrigerants and propellants, they are to as effective as CFC's and they tend to be more expensive Both HCF's and HCFC's are greenhouse gasses and contribute to the greenhouse effect. They should be collected and disposed of carefully rather than being released into he atmosphere. These compounds are a somewhat compromise between the environmental affects of CFC’s and their effectiveness as coolant and propellant. HCFC’s and HFC’s are a mediocre replacement for CFC’s, however more research will be required to find a group of chemicals which will act like CFC’s and have little to no ozone depletion potential and global warming potential.
Montreal Protocol
The Montreal Protocol on Substances that Deplete the Ozone Layer is an international treaty whose purpose is to protect the ozone layer by phasing out the production of numerous substances that are responsible for ozone depletion. They aim to reduce the impact of haloakanes on the ozone layer.
The following table highlights the measures, and target dates of the Montreal Protocol
The following table highlights the measures, and target dates of the Montreal Protocol
| Ozone depleting substances | Developed countries | Developing countries |
| Chlorofluorocarbons (CFCs) | Phased out end of 1995 | Total phase out by 2010 |
| Halons | Phased out end of 1993 | Total phase out by 2010 |
| CCl4(Carbon tetrachloride) | Phased out end of 1995 | Total phase out by 2010 |
| CH3CCl3 (Methyl chloroform) | Phased out end of 1995 | Total phase out by 2015 |
| Hydrochlorofluorocarbons (HCFC’s) | Freeze from beginning of 1996 | Freeze in 2013 at a base level calculated as the average of 2009 and 2010 consumption levels |
| 35% reduction by 2004 | 10% reduction by 2015 | |
| 75% reduction by 2010 | 35% reduction by 2020 | |
| 90% reduction by 2015 | 67.5% reduction by 2025 | |
| Total phase out by 2020 | Total phase out by 2030 | |
| Hydrobromofluorocarbons (HBFC’s) | Phased out end of 1995 | Phased out end of 1995 |
| Methyl bromide (CH3Br) | Freeze in 1995 at 1991 base level | Freeze in 2002 at average 1995-1998 base level |
| (Agricultural uses) | 25% reduction by 1999 | 20% reduction by 2005 |
| 50% reduction by 2001 | Total phase out by 2015 | |
| 70% reduction by 2003 | ||
| Total phase out by 2005 | ||
| Bromochloromethane (CH2BrCl) | Phase out by 2002 | Phase out by 2002 |
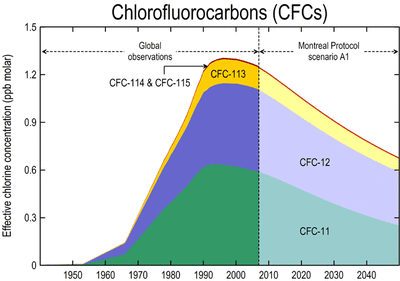
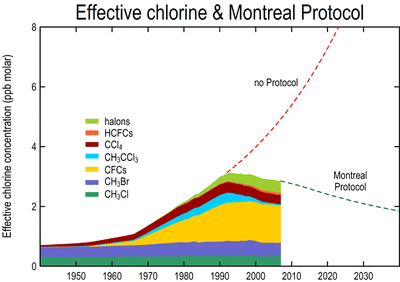
The Montreal protocol has reduced the quantity of chlorine released into the atmosphere and reduced the amount of CFC’s which are used, thus it has been an affective agreement as it is achieving what it originally aimed to do. This can be observed in the following graphs.
When studying the replacement chemicals (HFC's and HCFC's) for CFC's it is important to consider both the advantages and disadvantages of each
| HCFC’s | HFC’s | |
| Advantages | The suitability of the properties of replacement chemicals as to their application | HFC’s contain C-H bonds and as a result undergo some decomposition in the stratosphere, meaning they have zero effect on the ozone layer |
| The ozone depletion potential of the replacement chemicals, they | HFC’s contain no chlorine at all, so they have a zero ozone depletion potential, even if they do reach the stratosphere | |
| Disadvantages | A substantial proportion if intact molecule will reach the stratosphere where they have ODP | HFC’s are essentially greenhouse gasses and therefore contribute significantly to global warming |
| In the stratosphere, radiation can break C-Cl bonds leading to Cl free radical emission, starting a chain reaction which will slowly destroy the ozone layer, this is less, but still destructive | They absorb heat radiation from the earth’s surface at a much greater rate than carbon dioxide (the standard), some up to 10,000 times. | |
| Toxic, flammable, corrosive and explosive | ||
| GWP of 1000 times that of CO2 | ||
| Greenhouse gas, so has a high GWP | ||
| Costly |
CFC’s will persist in the atmosphere for some time, as there is already a high concentration of them in the troposphere. CFC’s must diffuse up into the stratosphere, however diffusion is a slow process and this could take some 50-100 years for all CFC’s to diffuse if further use of CFC’s is inhibited. Once in the stratosphere they will begin to undergo reactions to break down ozone, thus destroying the CFC molecule.
The Montreal protocol has significantly reduced the quantity of harmful substances, which act upon the ozone layer. Despite total ozone recovering needing 50-100 years, the quantities of chlorine and CFC’s have been greatly reduced to what it was 35 years ago. The release of harmful chlorine peaked in the 1990’s having released 3ppb molar of chlorine atoms into the atmosphere, and to date, this has been reduced to 2ppb molar and is expected to continue to reduce over the future. In 1995 CFC concentration peaked at 1.3ppb molar of CFC substances released into the atmosphere. Since the introduction of the Montreal Protocol in 2007, the quantity of atmospheric CFC’s have declined to 1.0ppb molar at present. Reducing the amount of chlorine and CFC’s which re released into the atmosphere, has hade a healing affect on the ozone layer, allowing for the ozone hole, and ozone depletion to slowly recover, however this is a long process as diffusion into the stratosphere takes time.